How Our GeneTAC® Molecules Work
Design is pioneering the field of small molecule genomic medicine, which holds the potential to safely and effectively deliver meaningful change to patients in need.
Our novel small molecule therapeutic candidates are GeneTAC® (Gene Targeted Chimera) molecules that target the underlying cause of inherited genetic diseases.
Through our proprietary GeneTAC® approach, we can design and develop therapeutic candidates that dial gene expression up or down without requiring gene editing.
Clinical results from our lead GeneTAC® small molecule (DT-216) for Friedreich ataxia have shown that our GeneTAC® approach can modulate target gene expression in patients after a single dose, demonstrating the mechanism and promise of this platform.
Singularly Rare, Collectively Common
Nucleotide repeat expansion mutations have been identified as the underlying cause of more than 40 debilitating, and typically rare, degenerative genetic diseases that collectively impact millions of people worldwide.
Individuals with nucleotide repeat expansion diseases are born with abnormally expanded stretches of specific nucleotide sequences, often with hundreds to thousands of repeats present in the mutant gene. Nucleotide repeat expansions are pathologic when they either impair transcription of the mutated gene (e.g., in Friedreich ataxia) or lead to formation of a pathogenic mRNA or mutant protein (e.g., Fuchs endothelial corneal dystrophy or Huntington disease). A higher number of excess repeats can lead to more severe, and sometimes a more rapidly progressive, form of disease.
Currently, there are no approved disease modifying therapeutic options that address the cause of nucleotide repeat expansion diseases.
An Elegant Solution
The development of drugs to treat genetic disease has largely focused on novel complex modalities such as gene therapy or gene editing that have proven to be challenging to develop. Until recently, scientists had not succeeded in using small molecules for selective, targeted transcription modulation. Through our GeneTAC® platform, we have designed small molecules that effectively target and dial up or down the expression of specific genes, with one of our lead compounds having entered the clinic in 2022. We have shown that GeneTAC® small molecules can directly distribute into multiple tissues and can be formulated for different routes of administration. Further, applying our substantial knowledge of medicinal chemistry to our GeneTAC® platform enables us to better design and select molecules with favorable pharmacokinetics, safety profiles, biodistribution, and biological activity, enabling a more straightforward and predictable translation into the clinic. Similar to other small molecules, GeneTAC® molecules can be tailored to achieve a desired effect without requiring cutting, editing or inserting exogenous genes, thereby avoiding the challenges and risks of developing a therapeutic that causes a permanent genetic change.
The GeneTAC® Molecule: A New Class
The structures of GeneTAC® molecules enable them to bind and act specifically at the site of the disease-causing nucleotide repeat expansion. They target the mutant gene and modulate the cell’s native transcriptional machinery, selectively halting or resuming the expression of specific genes and in turn blocking pathogenic mRNA or protein production and restoring functional protein production.
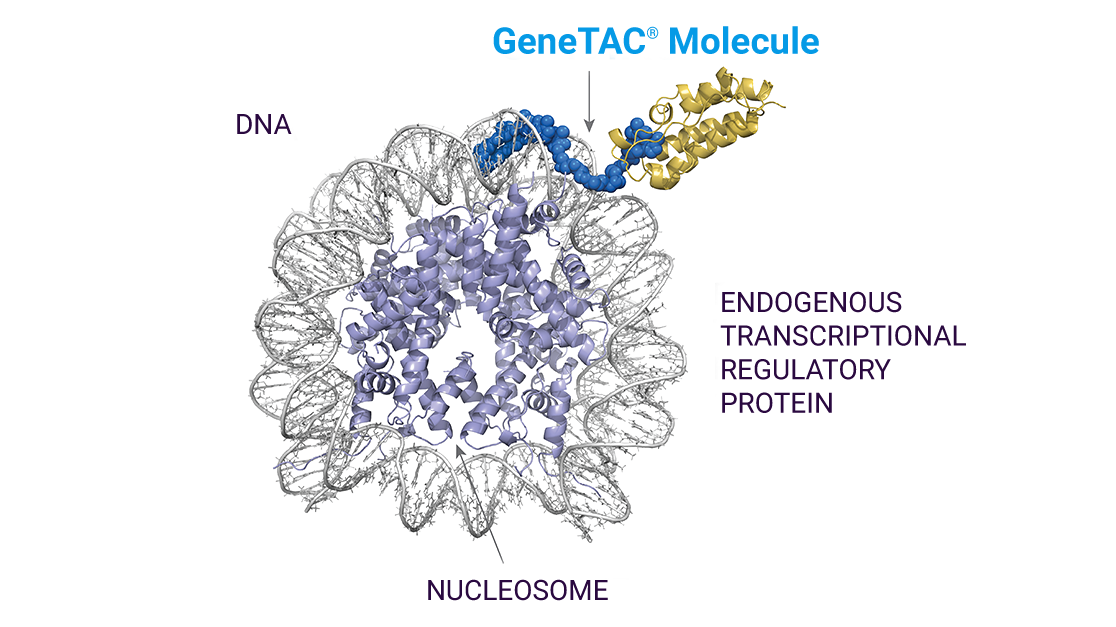
The versatility of the GeneTAC® platform has allowed us to design GeneTAC® molecules toward a specific nucleotide repeat expansion sequence on a specific mutant gene, regardless of the number of repeats, and tailor it to address the underlying disease-specific dysfunction, whether that requires restoration of transcription or reduction of toxic gene product levels.
